|
eCQM Solutions
|
Delivering an accurate electronic report to CMS for an Eligible Professional requires thorough review of a multi-stage review process:
CMSGATEWAYS.COM is a leader in
providing to software tools that streamline the
design and generation of custom CMS submissons. |
|||||
|
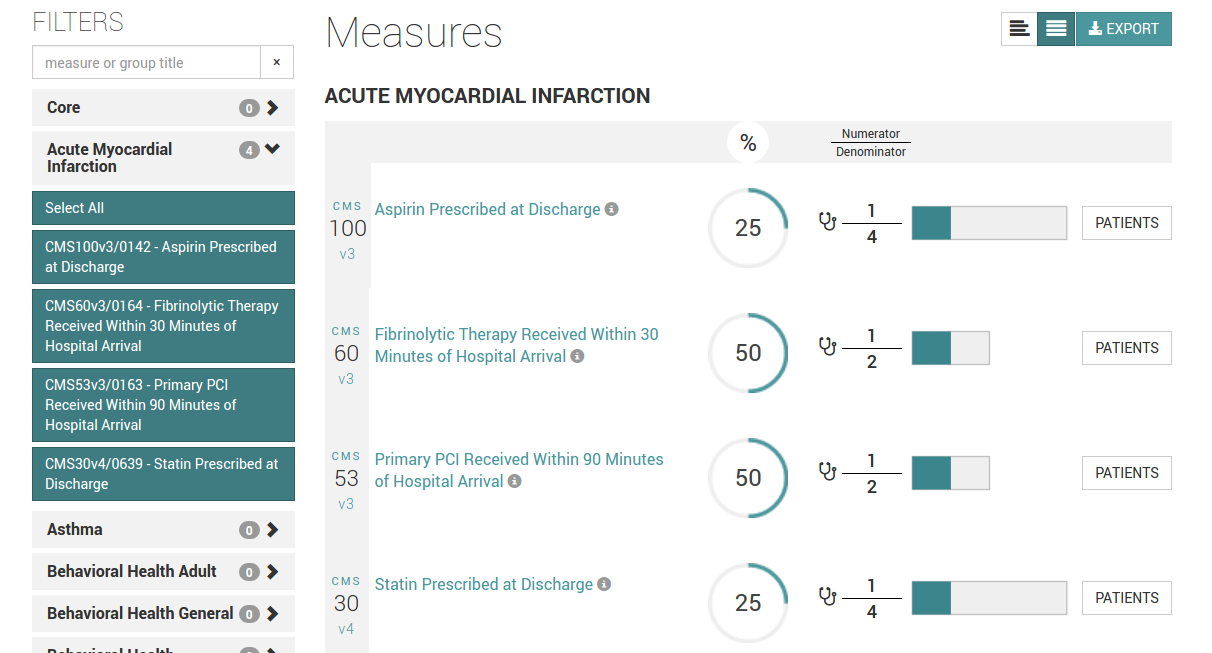
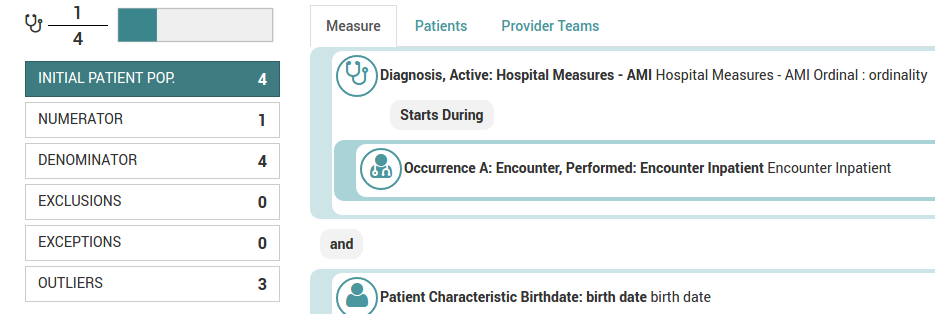
Our eCQM compilation is a three stage process. This is to accommodate many different data formats found in the inbound EHR datastreams.
Using this three stage system, eCQM reports can be compiled according to the capabilities of the respective EHR systems and merged into a common data stream. |
|||||